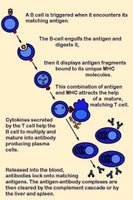
The
complement system comprises an assembly of liver-manufactured, soluble and cell-bound proteins that participate in
innate and
adaptive immunity.
Activation of the complement cascade by
protease cleavage leads to
chemotaxis (C5a),
inflammation and increased capillary permeability (C3a, C5a),
opsonization (C3b), and
cytolysis.
▼
activation :
alternative pathway :
amplification by C3 :
anaphylatoxins C3a,
C5a :
antigen uptake :
C1 :
C2 :
C3 :
C4 :
C5 :
C6 :
C7 :
C8 :
C9 :
C1INH :
CD59 :
chemotaxis :
classical pathway :
complement cascade :
complement control proteins :
convertases C3,
C3/C5,
C5 :
disorders :
evolution :
Factor B :
Factor H :
Factor I :
ficolins :
immunoglobulins and complement activation :
inhibitory proteins :
lectin pathway :
MBL -MAPS :
mannose-binding lectin pathway :
membrane attack complex :
opsonin :
pathways :
perforin :
phagocytosis :
pore :
proteases :
regulation :
serine proteases :
sialic acid ▼
Sequential
activation of the protein components of the
complement cascade upon cleavage by a
protease, leads to each component's becoming, in its turn, a
protease. Three pathways are involved in complement attack upon pathogens:
●
classical pathway●
alternative pathway●
mannose-binding lectin pathway (
MBL -MAPS)
The
classical pathway utilizes C1, which is activated by binding of an
antibody to its
cognate antigen.
Inactive
C1 circulates as a serum molecular complex comprising 6 C1q molecules, 2 C1r molecules, and 2 C1s molecules. Constant regions in some
immunoglobulins specifically bind C1q, activating C1r and C1s. The mu chains of IgM and some gamma chains of IgG contain specific binding sites, though IgM is far more effective than IgG.
Activated
C1s is a
serine protease that cleaves
C4 and
C2 into small inactive fragments (C4a, C2a) and larger active fragments,
C4b and
C2b. The active component C4b binds to the sugar moieties of surface glycoproteins and binds noncovalently to C2b, forming another
serine protease C4b•C2b, which is called
C3 convertase because it cleaves
C3, releasing an active C3b
opsonin fragment.
Macrophages and
neutrophils possess receptors for C3b, so cells coated with
C3b are targetted for
phagocytosis (
opsonization). The small
C3a fragment is released into solution where it can bind to
basophils and
mast cells, triggering histamine release and, as an
anaphylatoxin, potentially participating in anaphylaxis.
C3 amplifies the
humoral response because of its abundance and its ability to auto-activate (as a
C3 convertase). Breakdown of C3b generates an antigen-binding C3d fragment that enhances antigen uptake by
dendritic cells and
B cells .
Binding of
C3b to
C5 induces an allosteric change that exposes C3b•C5 to cleavage by C4b•C2b, which is now acting as
C3/C5 convertase. The
alternative pathway possesses a distinct
C5 convertase, so the two pathways converge through C5.
Cleavage of C5 by the
C3/C5 convertase releases:
●
anaphylotoxic C5a, which promotes
chemotaxis of
neutrophils● C5b, which complexes with one molecule of each of C6, C7, and C8. The resultant
C5b•6•7•8 complex assists polymerization of as many as 18
C9 molecules to form a cytolysis-promoting
pore (
membrane attack complex,
tem) through the
plasma membrane of the target cell, which then suffers osmosis-induced
cytolysis.
Another
cytolytic mediator utilized by
CTLs and
NK cells is
perforin, which is a 534 aa glycoprotein with sequence
homology to the
membrane attack component of
complement C9. Like C9,
perforin integrates into the target
cell membrane, forming polyprotein pores up to 20nm in diameter comprising 12—18 perforin monomers, which breach membrane integrity and permit cytolytic cell death.
The
alternative pathway is
not activated by antigen-antibody binding, but instead relies upon
spontaneous conversion of C3 to
C3b, which is rapidly
inactivated by its binding to
inhibitory proteins and
sialic acid on the cell's surface. Because bacteria and other foreign materials lack these inhibitory proteins and sialic acid, the C3b is not inactivated and it forms the C3b•Bb complex with
Factor B. The C3b.Bb complex acts as a
C3 convertase, forming C3b•Bb•C3b, which acts as a
C5 convertase that can ititiate assembly of the membrane attack complex. C3b•Bb, acting as a
C3 convertase, provides a positive feedback loop that amplifies production of C3.
The
lectin pathway (MBL - MASP) is homologous to the
classical pathway, but utilizes
opsonin,
mannan-binding lectin (
MBL,
MBP) and
ficolins rather than
C1q. Binding of mannan-binding lectin to mannose residues on the pathogen surface activates the
MBL-associated serine proteases, MASP-1, MASP-2, MASP-3, which cleave C4 into C4b and C2 into C2b. As in the classical pathway, C4b and C2b bind to form the C4b•C2b
C3 convertase.
Ficolins are homologous to MBL and function through MASPs. Diversified ficolins are of particular importance in invertebrates, which lack the adaptive immune response that
evolved some 500 million years ago in jawed vertebrates.
Several
complement control proteins regulate activity of the complement system, including:
● C1 inhibitor (C1INH), which eliminates the proteolytic activity of activated
C1r and
C1s. Following C1 activation by antigen-antibody complexes, C1INH permits only a brief interval during which activated C1 can cleave C4 and C2.
● Factor I, which inactivates
C3b● Factor H, which removes
Bb, thus interrupting the
C3 convertase feedback loop within the
alternative pathway● CD59, which inhibits
C9 polymerization during assembly of the
membrane attack complex.
Dysregulation of the complement system manifests variously as immune complex disorders (C2 deficiency), susceptibility to bacterial infections (C3 deficiency), the autoimmune disorder SLE (early component or C2 deficiency), hereditary angioneurotic edema (HANE) (C1INH deficiency).
▲
activation :
alternative pathway :
amplification by C3 :
anaphylotoxins C3a,
C5a ф
antibodies ф
antigen :
antigen uptake :
C1 :
C2 :
C3 :
C4 :
C5 :
C6 :
C7 :
C8 :
C9 :
C1INH :
CD59 :
chemotaxis :
classical pathway :
complement cascade :
complement control proteins :
convertases C3,
C3/C5,
C5 :
disorders :
evolution :
Factor B :
Factor H :
Factor I :
ficolins :
immunoglobulins and complement activation :
inhibitory proteins :
lectin pathway :
MBL -MAPS :
mannose-binding lectin pathway :
membrane attack complex :
opsonin :
pathways :
perforin :
phagocytosis :
pore :
proteases :
regulation :
serine proteases :
sialic acid ▲
Tables
Fc receptors
Immune Cytokines
Immunoglobulins▲
Top ▲
tags
[Immunology][complement]Labels: alternative pathway, anaphylatoxin, B cells, classical pathway, Clonal Ignorance, complement, complement cascade, dendritic cells, ficolin, mannose-binding lectin pathway, perforin, protease
|