T cells or T lymphocytes are specialized
lymphocytes distinguished by their
TCR surface receptors.
▼
activation T reg :
APC :
apoptosis :
B7-1 :
B7-2 :
CD8 + :
CD28 :
CD80 :
CD86 :
costimulation :
CTL :
cytotoxic T lymphocytes :
Fas-FasL :
γδ T cells :
granulysin :
granzyme :
helper T cell :
IL-2 :
interleukin-2 :
natural killer T cells :
NKT :
perforin :
pore :
self-protection :
serine protease :
Tc :
Th :
Treg :
Treg activation/action :
T cell types :
VDJ recombination ▼
'T' lymphocytes initially develop in the
thymus, and differentiate peripherally into several functional subsets of cells:
●
cytotoxic T cells –
CTL,
Tc –
CD8 +●
helper T cells, effector T cells –
Th –
CD4 +●
regulatory T cells –
Treg (formerly termed 'suppressor' T cells)
●
natural killer T cells –
NKT (different than
NK cells)
●
γδ T cells Cytotoxic T lymphocytes
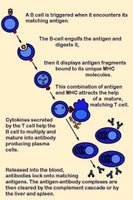
Cytotoxic T lymphocytes (CTLs) or
killer T cells express
antigen-specific,
VDJ recombination-generated
TCRs plus glycoprotein
CD8 on their
surfaces. The TCR recognizes specific antigenic proteins that are fragmented into peptides and then are
presented in complex with
MHC class I molecules (MHC-peptide complex - red).
APCs provide synergistic
signaling by
costimulatory molecules. APC signaling is necessary to sustain and integrate TCR signaling and to stimulate optimal T cell
proliferation and
differentiation.
The binding of TCR•antigen•CD8•MHC-I holds the
activated CTL (Tc) in proximity to the damaged/infected target cell. Once activated, the Tc cell undergoes clonal expansion with the assistance of a
cytokine, interleukin-2 (
IL-2) that acts as a
growth and
differentiation factor for T cells. (Table
Immune Cytokines)
CD28 binds the
CD80 (
B7-1) and
CD86 (
B7-2) ligands that are expressed on antigen presenting cells (
APCs). When exposed to infected/dysfunctional somatic cells, Tc cells release
perforin, which form pores in the target cell's
plasma membrane. Also released from Tcs are
granulysin and the
serine protease granzyme, which transit the
perforin pore and induce
apoptosis of the target cell.
A second
cytolytic mechanism is mediated by the interaction of activated T cell surface
FasL with
TNFR family
Fas receptors (
TNFRSF6, Apo_1, CD95) expressed on the surface of the target cell.
 Helper T cells
Helper T cells (
Th), or effector T cells circulate throughout the body where they interface with
MHC class II protein on other cells, determining whether the MHC class II is presenting 'self' or 'non-self protein' (
antigen). Th cells direct antibody class switching in B lymphocytes.
When the helper T cell is activated by contact with
antigen, it enters the
cell cycle in addition to producing
lymphokines and
chemokines. Th cells orchestrate
activation and growth of
cytotoxic T cells, and maximize the bactericidal activity of
phagocytes (
macrophages).
Helper T cells mostly carry the
CD4 surface protein, though a few carry
CD8. The
CD4 receptor triggers targetting by HIV, which determines the crippling effect of HIV on the immune system.
Regulatory cells (Treg)
Regulatory T cells were formerly called
suppressor T cells because they suppress
immune responses. Negative regulatory functions have been ascribed to several T cell types:
●
CD4+CD25+
●
CD4+CD25–
●
interleukin-10-producing TR1 cells
●
transforming growth factor-ß-secreting TH3 'regulatory' cells
[
v]
Treg cells express the αβ
TCR chains and are
activated only when the TCR binds to its cognate peptide-class
II MHC molecule
and receives
costimulation from
B7 molecules (
CD80,
CD86) on the
antigen-presenting cell. Once activated, Treg secrete
interleukin 9 (
IL-9),
interleukin 10 (
IL-10), +/-
transforming growth factor-beta (
TGF-β), and these
lymphokines inhibit Th1 assistance of cell-mediated immunity and inflammation, and
Th2 assistance of antibody production, and may inhibit the action of
CD8+ CTLs.
Because the antigenic peptides recognized by Treg TCRs are typically
self-peptides, the chief function of Treg cells is provision of self-protection by preventing other T cells from attacking self components, thus protecting against
autoimmunity.
CD25, or IL-2 receptor alpha chain, or
IL-2R, or Tac antigen is a 55kDa glycoprotein whose transcription is regulated by Stat5, Elf-1, HGG-I(Y), GATA, and Tax from HTLV-1. CD25 is expressed on PHA-stimulated T cells, B cells stimulated with anti-IgM antibody, monocytes/macrophages stimulated with LPS, and highly expressed on HTLV-I-transformed T cell lines. NCBI LocusLink Record:
3559Natural killer T cells – NKT cells are activated by group 2,
CD1d molecules to secrete interferon-γ (
IFNγ) and
IL-4, which act as
Th1 and
Th2 cytokines.
γδ T cells carry
TCRs encoded by different gene segments than those of αβ T cells.
CD27 is cell-surface marker for T-cell progenitors that are beginning to undergo commitment to the αβ or γδ T-cell lineages, and newly emerging αβ and γδ T-lineage cells display early differences in dependence on
Notch–Delta signaling and display distinct patterns of gene expression.[
npg]
Presentation of
peptide antigens for
activation of naïve T cells does
not reside solely in
dendritic cells. A population of γδ T cells can efficiently present peptide antigens to αβT cells, which comprise the predominant mammalian T cell population. Thus, γδ T cells function to bridge the
innate and
adaptive immune responses.
γδ T cells can form unique memory subsets with distinct
adhesion and
chemotactic capabilities, determining the
tissue homing capabilities of cells. γδ T cells are enriched in areas of
inflammation. Human γδ T cells express ligands for E- and P-
selectins, so they display specialized homing to epithelial surfaces. The memory γδ T cells express
chemokine family receptors not expressed by αβ T cells.
Human γδ T cells that express Vγ2Vδ2 TCRs can recognize
nonpeptide antigens and Non-Hodgkin's B cell lymphomas. In particular, recognition of prenyl pyrophosphates is performed by this extracellular presentation pathway, which is distinct from
MHC and
CD1 presentation, and which is important in many different
bacterial and parasitic infections. However,
γδ T cells of the major tissue subset recognize self and foreign nonpeptide antigens presented by CD1. These nonpeptide antigens are lipids and glycolipids presented by
CD1 proteins, which are MHC-like-molecules specialized for the presentation of lipids.[
s].
▲ф ф
antibodies ф
antigen :
APC ф
APCs :
apoptosis סּ
apoptosis :
B7-1 :
B7-2 ф
CD :
CD8 + :
CD28 :
CD80 :
CD86 סּ
cell growth סּ
cell membranes ₪
cellular fate :
costimulation ф
costimulation :
CTL ~
cytokines ф
cytolysis :
cytotoxic T lymphocytes ₪
differentiation :
Fas-FasL :
γδ T cells :
granulysin §
granulysin :
granzyme :
helper T cell ф
helper T cell :
IL-2 ~
immunoglobulins :
interleukin-2 ф
immune cytokines
Immune Cytokines :
killer T cells ф
killer T cells ф
lymphocytes ф
lymphokines ф
MHC :
natural killer T cells :
NKT :
perforin :
pore ф
receptors :
serine protease ф
signaling ф
surface receptors:
Tc :
Th :
Treg :
T cell types ф
thymus :
VDJ recombination ф
VDJ recombination ▲ф
Tables
Cell Adhesion Molecules
Cell signaling
Complement Receptors
Cytokines
Fc receptors
Immunoglobulins
Interferons
Receptor Tyrosine Kinases (RTKs)
Receptor Signal Transduction
Scavenger Receptors
Second Messengers
Toll-like Receptors▲
Top ▲
æ
Archaea & Eubacteria ¤
Cancer סּ
Cell Biology ~
Chemistry of Life
Diagrams & Tables ♦
Enzymes ₪
Evo Devo ○
Molecular Biology φ
Molecules ō
Organics ›››
Pathways ▫▫
Virus Labels: antigen presentation, APC, cytotoxic, helper, natural killer, Regulatory T cell, T lymphocytes, TCR, γδ T cells
|