anergy
Anergy toward self-targets operates as one self-tolerance mechanism to control the autoreactive cells found in autoimmunity. Clonal deletion in which lymphocytes are killed if they recognize a self-antigen during their maturation in the thymus gland or bone marrow is a major mechanism for the prevention of autoimmunity. However, not all human self-antigens are expressed in the central lymphoid organs where the lymphocytes are developing. Thus, self-tolerance to an individual's own antigens must also depend on mechanisms such as clonal anergy. Theoretically, recognition of a self-antigen eliminates the proliferative capacity of autoreactive lymphocytes in the peripheral immune system. Another process, immunoregulation, utilizes regulatory T cells that weaken harmful or inappropriate lymphocyte responses.
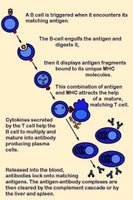
In B cell anergy, self-reactive B cells persist in the periphery yet remain unresponsive to immunogen. Research findings indicate that continuous binding of antigen and subsequent receptor signaling are essential for the maintenance of anergy.[n]
T cell anergy is induced when TCR stimulation "freezes" T cell responses until they receive an adequate subsequent antigenic signal from an antigen-presenting cell. Such APC signals can rescue T cells from anergy, stimulating them to produce the lymphokines necessary for the growth of additional T cells.
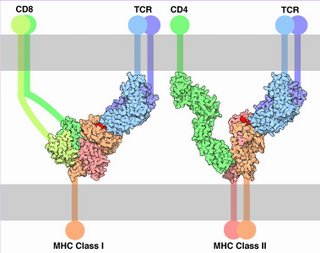
During a productive immune response, CD4+ T cells respond to effective signals by producing interleukin 2 (IL-2) and by proliferating. Effective signals stimulate require both ligation of TCRs with cognate antigens presented by class II MHC molecules on the surface of APCs and activation of costimulatory receptors, such as CD28, which recognize ligands such as B7 proteins expressed on the surface of APCs.
When T cells receive stimulus only TCR signals in the absence of engagement of costimulatory receptors, they enter a state of anergic unresponsiveness characterized by an inability to produce IL-2 or to proliferate upon re-stimulation. Such anergic T cells show a profound block in Ras/MAPK pathway that prevents activation of the AP-1 family of transcription factors (Fos/Jun).
GRAIL (gene related to anergy in lymphocytes) is GRAIL is an E3 ubiquitin ligase that is necessary for the induction of CD4+ T cell anergy in vivo. It is upregulated in naturally occurring (thymically derived) CD4+ and CD25+ cells [a] and anergized T cells [1]. Both GRAIL and Foxp3 are genotypic marker for CD25+ Treg cells. T cell activation appears to be controlled by Foxp3 through transcriptional regulation of early growth response (Egr) genes Egr-2 and Egr-3, and E3 ubiquitin (Ub) ligase genes Cblb [?], Itch [?] and GRAIL, subsequently affecting degradation of two key signaling proteins, PLCgamma1 and PKC-theta. [a]
It is believed that GRAIL could induce anergy through ubiquitylation of membrane-associated targets required for T-cell activation. It has been demonstrated that two isoforms of otubain-1, in conjunction with the deubiquitylating enzyme USP8, produce opposing effects on the expression and function of GRAIL in the induction of anergy.[2] GRAIL is differentially expressed in naturally occurring and peripherally induced CD25+ Treg cells where the expression of GRAIL has been suggested is linked to their functional "regulatory" activity.
Tables Complement Receptors Cytokines Fc receptors Immune Cytokines Immunoglobulins Interferons Scavenger Receptors Toll-like Receptors
tags [Immunology] [anergy]
Labels: activated B cells, affinity maturation, anergy, autoimmunity, B lymphocytes, BCR, bone marrow, CD28, CD4+, clonal deletion, GRAIL, lymphoid, lymphokines, MHC, self-tolerance, T cells, TCR, thymus
| 0 Guide-Glossary