Dendritic cells
Dendritic cells and their immature counterparts,
Langerhans cells (LC), are highly specialized, professional
antigen-presenting cells (APC) located in the skin, mucosa, and lymphoid tissues.
▼
adhesion :
APC activities :
clonal expansion B cells :
cytokines :
DC types :
disorders :
ectopic FDC-formation :
follicular dendritic cells (
FDC) :
FDC networks :
generating germinal centers :
germinal centers :
immature dendritic cells :
immune regulators :
immunological synapse :
interferon producing cells :
lymphoid dendritic cells :
maturation :
morphology :
myeloid dendritic cells :
pDC :
plasmacytoid dendritic cells (
PDC,
IPC) :
precursor dendritic cells :
regulators :
Th1 and Th2 stimulation :
TLRs :
types of DC :
veiled cells ▼
Immune dendritic cells are named for their
morphology (long surface projections), and bear no relationship to neurons. Immature dendritic cells are also called '
veiled cells' because they display large cytoplasmic 'veils' rather than dendrites. DC and LC play a key role in the induction phase of contact allergenicity.
As
key regulators of
immune responses,
dendritic cells (DC) stimulate
lymphocytes to perform
cell-mediated and
humoral immune responses against
pathogens and
tumor cells. DCs can also educate
T cells to tolerate
self-
antigens, thereby minimizing
autoimmune reactions.
Types of dendritic cell
●
follicular dendritic cells (origin?) –
FDC
●
lymphoid dendritic cells (
lymphopoiesis) –
●
myeloid dendritic cells (
monocytopoiesis) – MDC1, MDC2
●
plasmacytoid dendritic cells – PDC, IPC – the major producers of
type I interferon (
IFN)
Immature, precursor dendritic cells (
pDC) circulate throughout the body,
migrating to
lymphocyte rich tissues (such as spleen and lymph nodes) upon stimulating encounter with
antigen. The dendritic cells internalize the antigen, then digest, and externalize the fragmented antigen that they
present to lymphocytes in
MHC-peptide complexes, expressing markers that stimulate lymphocyte
activation. Dendritic cells are the most effective
antigen presenting cells.
Follicular dendritic cells stimulate differentiation of
B cells,
monocytopoietic lineages (pDC1) stimulate differentiation of
Th1 cells,
lymphopoietic dendritic cells (pDC2) induce differentiation of
Th2 cells.
Plasmacytoid cells produce
type 1 interferon (IFN-α, β, Ω) and can
mature into dendritic cells that link
innate and
adaptive immune responses.
A variety of
factors operate in antigen recognition and processing by immature (precursor) dendritic cells and in the
maturation of immature cells.
Toll-like receptors on the surfaces of precurson dendritic cells recognize microbial components and induce the
differentiation of dendritic cell precursors.
GM-CSF and
IL-4 stimulate the maturation of
monocytopoietic pDC1, while
IL-3 stimulates the differentiation of pDC2. The
transition to mature dendritic cells down-regulates those factors that were involved in antigen internalization, while up-regulating the expression of MHC, costimulatory molecules that participate in lymphocyte activation, adhesion molecules, and specific cytokines and chemokines.
Adhesion molecules enhance direct interactions between T cells and dendritic cells (
immunological synapse). Dendritic cell stimulation of formation of
Th1 and
Th2 cells appears to be regulated by negative feedback. Th1 production of interferon-γ blocks the
further stimulation of Th1 differentiation by DC1 cells. Th2 production of
IL-4 kills the dendritic cell precursors that contribute to Th2 cell creation. Thus, although IL-4 stimulates Th2 differentiation, the promotion of Th2 cell formation by DC2 cells does not appear to involve IL-4. Costimulatory receptors CD80 and CD86 expressed by
mature dendritic cells activate T cells in concert with the recognition of antigen/MHC by the T cell receptor. The secretion of
IL-12 by dendritic cells stimulates T cell responses, in particular the differentiation of Th1 cells, which produce
interferon-γ and other
inflammatory cytokines.
Follicular dendritic cells are stromal cells unique to primary and secondary
lymphoid follicles. FDCs express all three types of
complement receptors as well as
Ig-Fc receptors, through which antigen-antibody immune complexes are retained. FDCs present native antigens to
potential memory B cells, of which only those coated with high affinity B cell receptors (
BCR) are able to bind.
Recirculating resting B cells migrate
through the
FDC networks.
Antigen-activated B cells undergo
clonal expansion within the FDC networks in a T cell-dependent fashion, generating
germinal centers. Evidence suggests the presence of two types of dendritic cells within human germinal centers: (i) the
classic FDCs that express
DRC-1,
KiM4, and
7D6 antigens represent stromal cells; and (ii) the newly identified
CD3-CD4-CD11c- germinal center dendritic cells (GCDC) represent
hematopoietic cells that may be analogous to antigen-transporting cells of mice.
Within germinal centers,
B cells undergo
somatic hypermutation, positive and negative
clonal selection,
isotype switching and
differentiation into high-affinity
plasma cells and
memory B cells. Adhesion between FDCs and B cells is mediated by ICAM-1 (CD54)-LFA-1(CD11a) and VCAM-VLA-4. T cells may interact with FDCs in a
CD40/CD40-ligand-dependent fashion.
Ectopic FDC-formation is found in a number of
autoimmune diseases and/or
chronic inflammatory situations, suggesting that FDC development is not restricted to secondary lymphoid organs, but rather that local conditions drives a precursor cell type into FDC-maturation. The precursor of FDCs has presently not been identified, but data suggests a close relation to fibroblast-like cells. [
s] It was initially believed that
all DCs were of
myeloid origin until several recent studies demonstrated that
some DCs could also be efficiently generated from
lymphoid-restricted precursors. FDCs appear to be involved in the growth of follicular lymphomas and in the pathogenesis of HIV infection.[
pm]
Lymphoid dendritic cells are of
lymphopoietic origin, and IL-3 stimulates the differentiation of pDC2 cells into DC2 cells, which stimulates differentiation of Th2 cells, which secrete the
lymphokine interleukins 4, 5, 10, and 13. (
IL-4,
IL-5,
IL-10, IL-13)
Myeloid dendritic cells are of
monocytopoietic origin, and the maturation of precursor cells (pDC1) is stimulated by
GM-CSF, and
IL-4. Mature DC1 cells secrete interleukin 12 (
IL-12), which acts through the JAK-STAT pathway to induce Th1 cells to secrete
TNF-β (lymphotoxin) and
IFN-γ. MDC-1 is the more common subtype, and is a major stimulator of Th
1 cell differentiation. MDC-2 is rare, and may function in response to wound infection.
Plasmacytoid dendritic cells (pDC=IPC) are the major producers of type I
interferon (
IFN) and exhibit the unique ability to link
innate and
adaptive immune responses, by
differentiating into DC capable of stimulating naive T cells and modulating the adaptive immune response. Human plasmacytoid DCs (PDCs) can induce either Th1- or Th2-type immune responses upon exposure to viruses or IL-3, respectively.
Plasmacytoid dendritic cell
precursors (
pDC) are type 1 interferon-(α, β, Ω)-producing cells (IPCs) that comprise 0.2%-0.8% of peripheral blood mononuclear cells (humans, mice). IPCs display
plasma cell morphology, selectively express
Toll-like receptor (TLR)-7 and TLR9, and rapidly secrete massive amounts of type 1 interferon following viral stimulation. IPCs promote the function of
natural killer cells,
B cells,
T cells, and
myeloid DCs through type 1 interferons (IFN) during an antiviral immune response. Later in
viral infection, IPCs differentiate into a unique type of mature dendritic cell, which directly regulates the function of T cells and thus links
innate and
adaptive immune responses. [
s,
↓] [
fft]
images []
sem dendritic cell and T cell []
micrograph Langerhans cells []
PKC bII signaling in dendritic cells []
micrograph gallery dendritic cells []
photomicrograph dendritic cells interacting with yeast (lilac) []
photomicrograph Human Dendritic cell (labelled with anti MHC class-I FITC) presenting Influenza antigens to T-lymphocytes.
videos Џ
Nature video library index Q Џ
ф
activation ф
affinity maturation ф
anergy ф
antibodies ф
antigen ф
APCs ф
autoimmunity ф
B cells ф
blood ọ
bone marrow ф
CD ф
cellular response ф
class-switch recombination ф
clonal selection ф
complement system ф
costimulation ф
helper T cell ф
hematopoiesis ф
humoral immunity ф
immune cytokines ф
immune response ф
immune tolerance ф
inflammatory response ф
interferons ф
isotype switching ф
killer T cells ф
lymphocytes ф
lymphokines ф
lymphoid system ф
lymphopoiesis ф
macrophages ф
MHC ф
monocytopoiesis ф
pattern-recognition receptors ф
phagocyte ф
plasma cells ф
receptors ф
signaling ф
somatic hypermutation ф
surface receptors ф
T cells ф
thymusTables
Complement Receptors
Cytokines
Fc receptors
Immune Cytokines
Immunoglobulins
Interferons
Scavenger Receptors
Toll-like ReceptorsIPC: professional type 1 interferon-producing cells and plasmacytoid dendritic cell precursors. [Annu Rev Immunol. 2005]
Plasmacytoid dendritic cell precursors/type I interferon-producing cells sense viral infection by Toll-like receptor (TLR) 7 and TLR9. [Springer Semin Immunopathol. 2005] PMID: 15592841 [
Free Full Text]
Natural type I interferon-producing cells as a link between innate and adaptive immunity. [Hum Immunol. 2002] PMID: 12480256
Thrombopoietin cooperates with FLT3-ligand in the generation of plasmacytoid dendritic cell precursors from human hematopoietic progenitors. [Blood. 2004] PMID: 14670916
Flexibility of mouse classical and plasmacytoid-derived dendritic cells in directing T helper type 1 and 2 cell development: dependency on antigen dose and differential toll-like receptor ligation. [J Exp Med. 2003] PMID: 12515817
Roles of toll-like receptors in natural interferon-producing cells as sensors in immune surveillance. [Hum Immunol. 2002]
tags
[Immunology] [dendritic+cell] [APC] [cytokine] [Toll-like+receptor]Labels: antigen presenting cells, APC, cytokines, dendritic cells, FDC MDC, interferon, IPC, Langerhans cells, LC, PDC, plamacytoid cells, Toll-like receptors, veiled cells
|


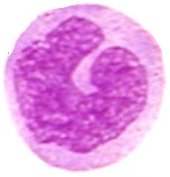
 Basophils are granulocytes packed with granules that stain basic (purple with H&E). The nucleus is bilobed, and the metachromatic granules contain sulfated glycosaminoglycans as well as vasoactive compounds – histamine and proteoglycans. Lysosomal arylsulfatase is found in granules of developing basophils in bone marrow. Basophil granules are surrounded by a unit membrane and contain particles which are uniform in size across the same granule yet vary in size in different granules within the same cell. Some granules reveal a homogeneous texture and/or "myelin" figures. [s] Though basophils and mast cells share morphological features, the appearance of most basophil granules differs from the ultrastructure of human mast cell granules.
Basophils are granulocytes packed with granules that stain basic (purple with H&E). The nucleus is bilobed, and the metachromatic granules contain sulfated glycosaminoglycans as well as vasoactive compounds – histamine and proteoglycans. Lysosomal arylsulfatase is found in granules of developing basophils in bone marrow. Basophil granules are surrounded by a unit membrane and contain particles which are uniform in size across the same granule yet vary in size in different granules within the same cell. Some granules reveal a homogeneous texture and/or "myelin" figures. [s] Though basophils and mast cells share morphological features, the appearance of most basophil granules differs from the ultrastructure of human mast cell granules.